 
This region also had the most end-to-end capability to translate vaccines from R&D through to manufacture and supplied products. 
The fourth region surveyed, South East Asia/ Western Pacific, showed more established manufacturing capabilities, with over 70% of respondents currently having planned capacity to produce over 50 million doses of vaccines this year (classifying them as having medium- or large-scale vaccine manufacturing capacity). By comparison, the US and Europe combined are expecting to produce several billion vaccine doses by the end of this year. For example, half of the vaccine production sites across Africa, Latin America, and the Middle East, combined, were expected to produce under 50 million vaccine doses for 2021. While current maximum production capacity across respondents for 2021 ranged from a few thousand (for clinical trials) to 800 million vaccine doses (for global commercial supply), this varied considerably across the regions surveyed. These key geographies are predominantly thought to be where vaccine production capabilities and capacities need to be established and/or expanded.Īvailable data, submitted by over 95 vaccine manufacturers, global health organisations, research and veterinary institutes, and government agencies from 37 countries across all four regions, from March to June 2021, confirmed this perspective. The study focussed on gathering data on vaccine manufacturing in Africa, Southeast Asia and the Western Pacific, the Middle East, and Latin America and the Caribbean. To know where and how we need to ramp up manufacturing capacity and capabilities, CEPI ran a first-of-its-kind landscaping exercise earlier this year to map all current and potential vaccine production efforts. Gathering the data on current and future vaccine production Boosting local production efforts would enable low- and middle-income countries to take full ownership of their national health security and accelerate distribution of vaccines and other countermeasures to their own populations. This has meant these regions have become dependent on the shipment of COVID-19 vaccines produced in other countries to innoculate their own populations – and they cannot always guarantee they will be first in line to receive these urgently needed doses.Īs part of our $3.5bn plan to build on these lessons and substantially reduce epidemic and pandemic risk, CEPI has set the goal to support the creation a globally distributed vaccine manufacturing network ready for future threats. 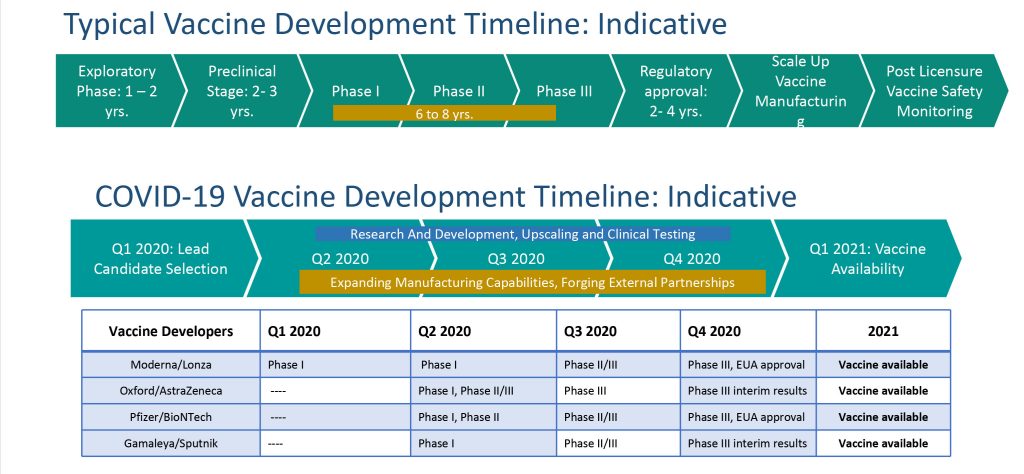
It has also exposed the ways that it can be improved to optimally support public health in the future.įor example, few nations currently have the end-to-end capacity to translate basic research into vaccine products within their borders as they lack the manufacturing capacity to scale-up production for large-scale testing and distribution. In fact, thanks to the triumphant work of manufacturers working around the clock, a historic 11 billion doses of COVID-19 vaccine are anticipated to be produced by the end of 2021-tripling previous annual vaccine output.Īt the same time, however, the COVID-19 pandemic has shone a light on some of the challenges within vaccine manufacturing and the overall vaccine ecosystem. Rapid vaccine production has been essential to the speedy global rollout of billions of doses of COVID-19 vaccines this year. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
